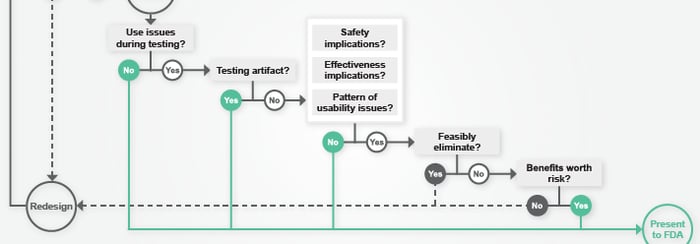
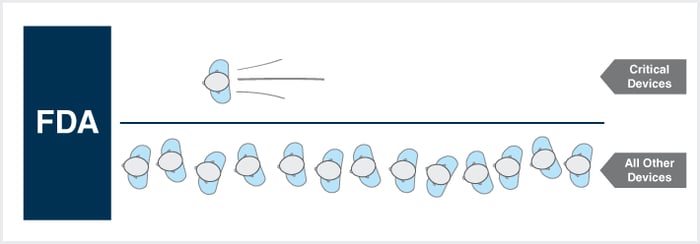
Over the past several years, there has been a drastic increase in the deployment of “interconnected” medical devices (e.g., wireless glucose monitors, infusion pumps, pacemakers, etc.). As our medical devices become more interconnected, they become increasingly critical to clinical decision-making and patient care. Access to information increases, decision making evolves, and, hopefully, patient. Read more
Steve Wilcox Serving as Include 2015 Conference Committee Chair
/ in conference , design , Helen Hamlyn , Include , Include 2015 , news , presentations , RCA / by Christina SInclude 2015, this year’s 8th International Conference on Inclusive Design, will involve a familiar face. Stephen Wilcox, founder and principal of Design Science, is serving as Chair of the Include Conference Committee. The biennial conference will be hosted by the Helen Hamyln Center for Design at the Royal College of Art (RCA). Read more
Design Science (San Bruno) to Host UX Meetup
/ in san bruno , san francisco , user experience , meetup , news , ux , ux speed dating / by Christina SOn July 15, Design Science will welcome Bay Area tech professionals to its office for the "UX Speed Dating" monthly meetup. This event provides web and mobile app designers a chance to gather in-person feedback from actual users.In a "speed dating with a twist" format, product owners sit stationary and administer their usability tests while product users rotate to a new station every 20 minutes.. Read more
Hospital, Home, and Beyond
/ in user-centered design , consumer insights , consumer products , contextual inquiry , customer education , ethnographic research , ethnography , news , personas , product design / by Christina SWhile Design Science is most well known for its experience in conducting ethnographic research at medical facilities, a growing number of our projects provide opportunities for us to venture outside of the hospital and apply our methodologies to the study of non-medical products. As a field researcher I have had the chance to participate in a number of “unconventional” projects that took me to. Read more
MDO Article: "Achieving Realism In Human Factors Work: How To Stay Out Of Fantasy Land"
/ in usability testing , FDA , healthcare , human factors , MDO , meddeviceonline , news / by Christina SDesigning usability studies that are "representative" is not only necessary for valid data, it's also an FDA requirement. Chad Uy shares some methods for maintaining realism to ensure representative usability studies in his recent article for Med Device Online. Read more
MDO Article: “How To Know You've Passed Validation Testing (And What To Do If You Haven't)”
/ in usability testing , design science , FDA , human factors , MDO , meddeviceonline , medical product design , news , presentations / by Christina SUnderstanding the ramifications of less-than-perfect validation testing results can be complex. In his recent article for Med Device Online, Peter Sneeringer presents 6 questions to help companies decide if errors seen during testing indicate a likelihood that the FDA will reject their submission. Read more
FDA to Accept Applications for Expedited Medical Device Approval
/ in usability testing , user-centered design , design science , EAP , Expedited Access PMA , FDA , guidance , healthcare , human factors , industrial design , medical product design , news / by Christina SOn April 15th, the Food and Drug Administration will begin accepting applicants through its new Expedited Access Premarket Approval Application for Unmet Medical Needs for Life Threatening or Irreversibly Debilitating Diseases or Conditions (“Expedited Access PMA” or “EAP”). Read more
MDO Article: "4 Proposals To Accelerate The Growth of mHealth"
/ in usability , user-centered design , app design , design science , healthcare , human factors , interaction design , MDO , meddeviceonline , mHealth , mobile app , news / by Christina SWhat 4 things should app developers, traditional medical device and pharmaceutical companies, and regulators do to accelerate the growth of mHealth?Check out Nicholas Araujo's proposals, featured on Med Device Online's website. Read more
Now Hiring: Research Associates
/ in usability testing , user-centered design , career , design science , ethnographic research , healthcare , human factors , industrial design , job opportunity , medical product design , news / by Christina SAre you passionate about working collaboratively to make a felt difference in the world? At Design Science we have one mission - fitting products to people. Read more
Design Internship Opportunity
/ in career , design science , industrial design , information design , interaction design , job opportunity , news / by Christina SAre you a designer with a passion for user research? Design Science is seeking an intern with a background in industrial or graphic design to work in our information design department. Read more